Blog
Featured Article
Portable Mixers with Pneumatic Drive: Handheld Pneumatic Mixers
Mark Hennis - President of INDCO, IncRecent Articles

Portable Mixing Solutions: 5 Gallon Bucket Mixers and More
Mark Hennis - President of INDCO, Inc
INDCO Mixes Efficiency for the Homebuilding Industry
Mark Hennis - President of INDCO, inc. Industries We Serve

Industrial Mixing Applications: Understanding the Difference between High Shear and Low Shear
Mark Hennis - President of INDCO, inc.
304 vs 316 Stainless Steel: What’s the Difference?
Mark Hennis – President of INDCO, Inc.
Difference between impellers and propellers in mixing
Mark Hennis - President of INDCO, inc.
How to Operate an Electric High-Speed Disperser Mixer: A Basic Guide
Mark Hennis - President of INDCO, inc.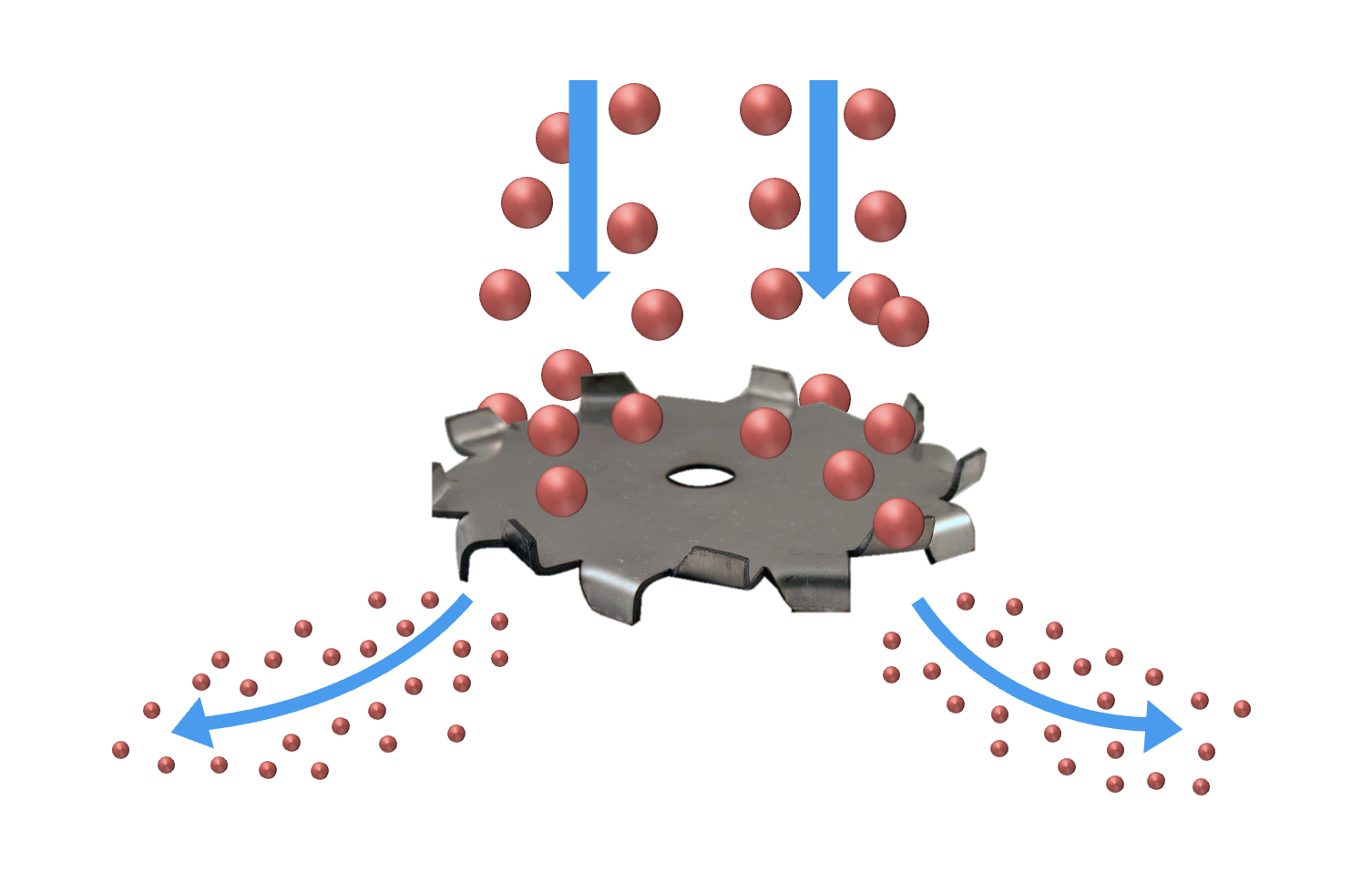
The Fundamentals of Dispersion in Mixing
Mark Hennis - President of INDCO, inc.








